c-Kit Mutation Detection Kit
Cat Number: CKIT-RT44
The c-Kit Mutation Detection Kit is a PCR-based assay that detects somatic mutations in the c-KIT gene, which are associated with tumor development, prognosis, and response to targeted therapies in cancers such as GIST, melanoma, and mastocytosis.
- Below 5% Limit of Detection
- 44 tests per kit
- 2-hour turnaround time
- Works with FFPE, fresh frozen and FNA samples
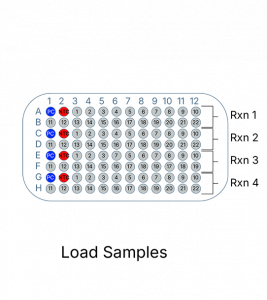
- 4 Reactions per sample
- For research use (RUO) in the U.S
- For in vitro diagnostic use (IVD) in the European Union
Mutations in the c-KIT gene are key drivers of tumorigenesis in various cancers, including gastrointestinal stromal tumors (GISTs), melanoma, and mastocytosis. These mutations lead to constitutive activation of the KIT receptor tyrosine kinase, promoting uncontrolled cell growth and survival.
The c-Kit Mutation Detection Kit is a real-time PCR-based assay designed for the rapid and sensitive detection of somatic mutations in the c-KIT gene. Using fluorescent hydrolysis probes, the assay accurately identifies mutant sequences even in samples with a high proportion of wild-type DNA.
It includes an internal control gene to ensure assay reliability and is optimized for use with FFPE and fresh/frozen tissue samples.
The c-Kit Mutation Detection Kit detects the following mutations:
| Mutation | COSMIC ID | Primer Mix Tube |
| EX11_p.V559D | COSM1252 | 1 |
| EX11_p.V559G | COSM1253 | |
| EX11_p.V560D | COSM1257 | |
| EX11_W557R | COSM1216 | |
| EX17_p.D816H | COSM1311 | |
| EX17_p.D816V | COSM1314 | |
| EX11_p.L576P | COSM1290 | 2 |
| 2EX13_p.K642E | COSM1304 | |
| EX9_p.Y503_F504insAY | COSM12444 | |
| EX11_p.W557_E561del | COSM18890 | 3 |
| EX11_p.W557_K558del | COSM12449 | |
| EX11_p.W557_V559>C | COSM1233 | |
| EX11_p.W557_V559>F | COSM1226 | |
| EX17_p.N822Y | COSM19109 | |
| EX17_p.K818R | COSM1315 | |
| EX17_p.N822H | COSM1318 | |
| EX11_p.V559A | COSM1255 | 4 |
| EX11_p.W557G | COSM1221 | |
| EX17_p.D820G | COSM1316 | |
| EX17_p.D820Y | COSM12710 | |
| EX17_p.N822K | COSM1322 |
For More Information
The c-KIT Mutation Detection Kit follows a simple and easy to use process. It can be completed in 2 hours and follows a simple straightforward process.
EntroGenʼs c-KIT Mutation Gene Detection Kit requires a real-time PCR instrument capable of detecting FAM, ROX and CY5 fluorescent probes. This test includes reagents required for PCR amplification/detection, as well as validated reaction controls.
Columns and reagents for DNA isolation are not included. Genomic DNA from FFPE tissues can be extracted using EntroGen pureNA® Genomic DNA Isolation Kit.




Click to download lot-specific quality control data for your product:







