ctDNA EGFR Mutation Detection Kit
Cat Number: ctEGFR-48
The ctDNA EGFR Mutation Detection Kit is a real-time PCR-based assay that detects somatic EGFR mutations in exons 18, 19, 20, and 21 from circulating tumor DNA, providing actionable insights for targeted therapy in lung cancer when tumor tissue is unavailable.
- 48 tests per kit
- 2-hour turnaround time
- 7 Reactions per sample
- For research use (RUO) in the U.S
- For in vitro diagnostic use (IVD) in the European Union
The epidermal growth factor receptor (EGFR) is a membrane protein that plays a central role in transmitting signals from extracellular ligands to intracellular pathways, such as the Ras-Raf-MAPK cascade, which drive cell proliferation and survival. Mutations in EGFR, particularly in exons 18, 19, 20, and 21, are frequently found in lung cancer and other solid tumors and have been shown to predict response to EGFR-targeted therapies.
The ctDNA EGFR Mutation Detection Kit is an ultra-sensitive, real-time PCR-based assay designed for the specific and sensitive detection of somatic EGFR mutations from circulating tumor DNA (ctDNA) isolated from plasma. Using mutant-specific primers and fluorescent hydrolysis probes, the assay preferentially amplifies mutant sequences even in samples with a high proportion of wild-type DNA.
This highly reliable and efficient assay provides valuable insights into tumor genetics.
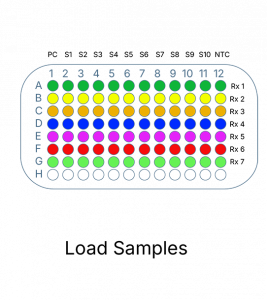
A summary of mutations detected in each reaction is shown in the table below:
| Reaction | Mutation |
| 1 | Exon 19 Deletions |
| T790M | |
| L858R | |
| 2 | C797S |
| 3 | L861Q |
| 4 | S768I |
| 5 | G719X |
| 6 | Ex20InsGGT/CAC |
| 7 | Ex20Ins9 (c.2300_2308dup) |
For More Information
The ctDNA EGFR Mutation Detection Kit follows a simple and easy to use process. It can be completed in 2 hours and follows a simple straightforward process.
EntroGenʼs ctDNA EGFR Mutation Detection Kit requires a real-time PCR instrument capable of detecting VIC, FAM, ROX, and CY5 fluorescent probes.
This test includes reagents required for PCR amplification/detection, as well as validated reaction controls.
Columns and reagents for cell-free DNA isolation are not included. Cell-free DNA can be extracted using EntroGen’s Cell-Free DNA Isolation Kit.




Click to download lot-specific quality control data for your product: