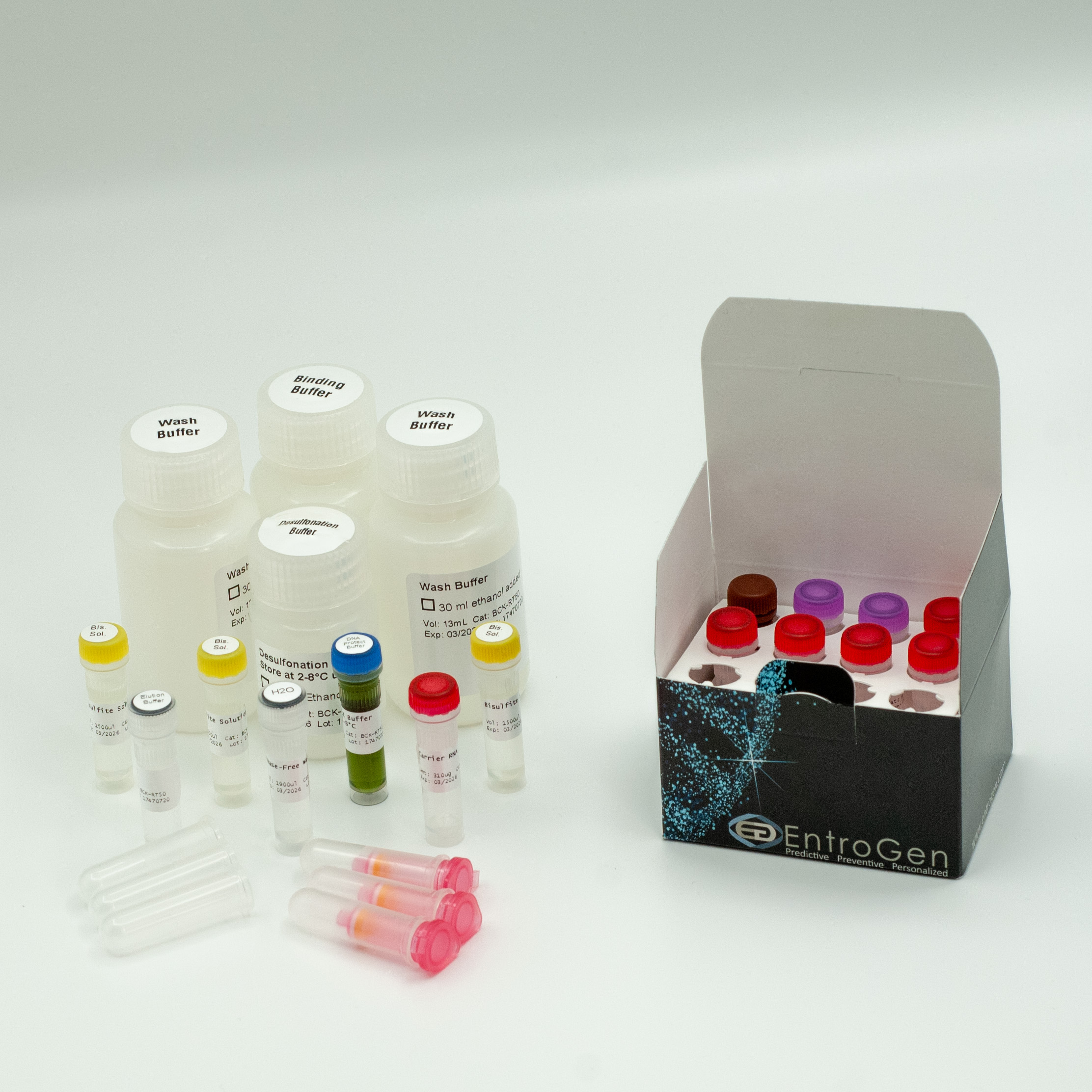
MGMT Methylation Detection Kit w/ Bisulfite Conversion Kit
Cat Number: MGMTX-RT44
The MGMT Methylation Detection Kit is a real-time PCR assay that detects promoter methylation in the DMR2 region of the MGMT gene using bisulfite-converted DNA, providing a sensitive method for assessing epigenetic silencing associated with treatment response in glioblastoma and other tumors.
- 60 Reactions per kit
- 3 Reactions per sample
- Bisulfite conversion kit is included
- Works with FFPE or fresh frozen samples
- 5% methylation reference control available
- For research use (RUO) in the U.S
- For in vitro diagnostic use (IVD) in the European Union
The MGMT Methylation Detection Kit with Bisulfite Conversion Kit is a semi-quantitative, multiplexed, real-time PCR-based assay that detects methylation in the MGMT DMR2 region using bisulfite-treated DNA.
The assay includes specific primers and fluorescent probes that selectively amplify and detect methylated cytosines, alongside an internal control gene (Actin B) for normalization and quality control.
This comprehensive kit provides all necessary reagents for efficient bisulfite conversion and methylation detection.
The MGMT Methylation Detection Kit utilizes the 5% Methylation Reference Control (MGMT-LPC) as a benchmark during the bisulfite conversion process. This control standard allows for the accurate calculation of process-corrected % methylation in unknown samples by accounting for run-to-run variations of the bisulfite reaction efficiency.
Researchers and clinical labs can reliably assess MGMT methylation status from FFPE or fresh/frozen tumor samples to support treatment decisions and prognostic evaluation.
The MGMT Methylation Detection Kit with Bisulfite Conversion offers a straightforward, streamlined workflow that can be completed in under a day, with minimal hands-on time and easy-to-follow steps.
EntroGen’s MGMT Methylation Detection Kit requires a real-time PCR instrument capable of detecting FAM and VIC fluorescent probes.
This test includes all necessary reagents for both bisulfite conversion and real-time PCR amplification/detection, along with validated controls. DNA is first extracted from FFPE or fresh/frozen samples, then subjected to bisulfite treatment using the included reagents to convert unmethylated cytosines into uracil. The treated DNA is then amplified using methylation-specific primers and fluorescent probes that target the MGMT DMR2 region and an internal control gene.
Columns and reagents for initial DNA extraction are not included in the kit. Genomic DNA from FFPE tissues can be extracted using EntroGen pureNA® Genomic DNA Isolation Kit.






Click to download lot-specific quality control data for your product: